Recombinant protein production entails manipulating the native protein expression to express proteins that do not occur in nature in large quantities for commercial use. The first recorded use of recombinant DNA technology was in the early 1980s when scientists made recombinant human insulin. Since then, recombinant protein production has gone mainstream, with the government records showing over 239 FDA-approved therapeutic proteins, with more in the clinical phase.
Besides therapeutics and medicine, recombinant proteins are also helpful in agricultural and nutritional research and industrial applications like cosmetics and detergent production. Below is a step-by-step break of how a recombinant protein production service works.
Obtaining A cDNA and Amplifying The Gene of Interest
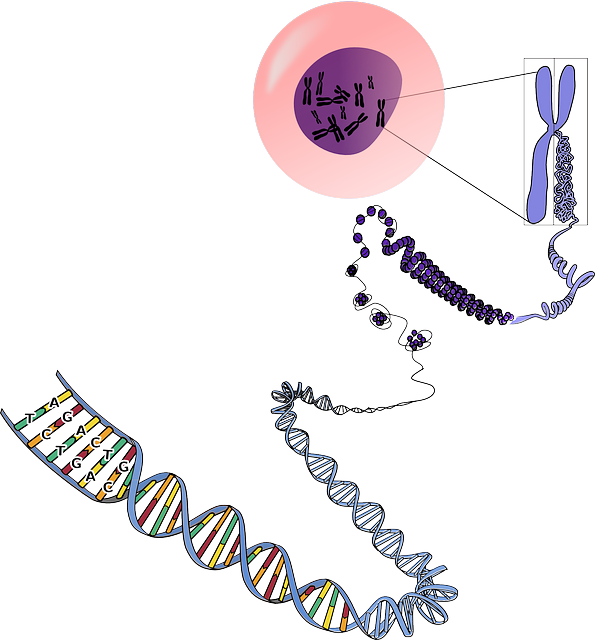
Native and recombinant protein production have one thing in common: they begin with a specific DNA sequence. DNA holds the code of instructions (genes) used in creating specific proteins and must transfer the set of instructions onto a template to initiate protein production. It does so utilizing two processes, transcription and translation.
The genetic information necessary for making the target protein molecule gets transferred onto a messenger RNA (mRNA) during transcription. An mRNA is a single strand of the DNA double helix and serves as a template for one of the base pairs in the target protein. The translation process comes after transcription and entails copying the mRNA sequence into amino acids that function as building blocks for the recombinant protein.
However, as stated above, most recombinant proteins feature gene sequences that do not occur naturally. Therefore, recombinant protein production begins with a DNA sample containing genetic material from various sources.
A restriction digestion enzyme gets introduced into the DNA sample to splice targeted genes and piece the spliced DNA strands in the sample using a ligand, facilitating a DNA clone formation. The transcription and translation processes occur, resulting in an mRNA formation.
Once the mRNA strand is ready, an enzyme called reverse transcriptase is added to the sample, facilitating complementary RNA (cRNQA) synthesis. A cRNA is an ideal medium for recombinant protein production because it contains the gene of interest.
However, gene amplification is necessary before proceeding to clone the realized cRNA. Gene amplification entails utilizing a process called polymerase chain reaction (PCR) to increase the target gene’s volume and, by extension, the number of recombinant proteins produced.

mRNA Cloning Into An Expression Vector
cRNA formation and amplification help generate the set of instructions necessary to translate a target protein. However, the DNA clone relies on an expression vector to facilitate recombinant protein expression.
An expression vector is a plasmid or a DNA molecule separate from the chromosomal DNA in a cell and replicates independently of the chromosomal DNA. Expression vectors help insert or introduce the DNA clone into a target cell or expression system for replication.
Plasmids typically feature a cylindrical shape with multiple functional sites, including restriction sites and multiple cloning sites. They are also biologically flexible and allow scientists to insert desirable genes and restriction sites.
Inserting desirable genes into a plasmid’s cloning site requires restriction enzymes that recognize restriction sites, cut out the present gene and insert the desirable gene. Since a multiple cloning site features various restriction sites, it is possible to clone an entire DNA sequence along with a multiple cloning site and bind the sequenced genes via DNA ligase.
Plasmids used as cloning vectors include bacteriophage or bacterial viruses that can replicate when inserted into a host cell. However, recombinant protein production also utilizes artificial chromosomes and cosmids.
Choosing The Ideal Protein Expression System
Cloning the mRNA into the cloning vector is part of the preparation process for the actual protein expression. Recombinant protein expression or synthesis occurs in a protein expression system, and below is a list of the protein expression systems utilized during recombinant protein production.
- Mammalian expression systems
- Insect expression systems
- Bacterial Expression systems
- Yeast expression system
- Algal expression systems
- Cell-free expression systems
Each expression system has advantages and disadvantages regarding speed, complexity, labor intensity and expertise, scalability, and cost. Second, the chosen expression vector must support protein expression within the selected expression system. Therefore, recombinant technology companies will recommend the ideal expression system for your target protein and its application.

Small-scale Test To Establish The Expression of The Target Protein
Although the steps above seem straightforward, protein expression is complex and may not go as planned. Therefore, a small-scale expression test is necessary to estimate the quantity of the target recombinant protein that the expression system can produce on a larger scale.
Such tests are conducted in a test tube and utilize target protein physical characteristics like solubility. Besides physical attributes, other methods like the northern blot to identify gene expression levels and fluorescent tagging are also helpful. Small-scale expression tests help researchers determine the course of action if the chosen expression system proves inefficient.
Protein Purification
Recombinant proteins expressed using various expression systems exist alongside other biological matter like cell debris and non-desirable proteins. Therefore, protein purification helps set the target protein apart, awaiting protein characterization.
The purification process also leverages the target protein’s physical characteristics, facilitating purification techniques like ion exchange, affinity chromatography, column chromatography, and electrophoresis. Each protein purification method has its advantages and disadvantages. Moreover, recombinant technology companies offer different packages for different protein purification levels.

Protein Characterization
While protein purification entails separating the recombinant proteins from physical impurities, protein characterization entails testing the proteins’ structure and functionality. Protein characterization ensures that the recombinant protein function has structural integrity and function as expected.
Conclusion:
Recombinant protein production was once exclusive to biotechnology experts, but amateur scientists can now offer the service. Moreover, more industries currently rely on recombinant technology to facilitate research and industrial product production. Therefore, understanding how the recombinant technology industry works helps when you need to acquire such services.